Last time, when talking about quantum mechanics, we used Hydrogen as our prime example.
That’s because Schrödinger’s equation for H can be exactly solved, but that’s not so for the other elements on the periodic table.
Any atom that contains two or more electrons is called a many-electron atom. This excludes H, He+, Li2+, etc.
Let’s jump into the many-electron atom!

We have found that the orbitals for a many-electron atom are very similar to those for the hydrogen atom – and the same quantum numbers, n, l and ml can be used to describe the orbitals in a many-electron atom. This approximation, known as the orbital approximation, converts a many-electron problem to many one-electron problems.
The energy levels for the many-electron atom are quite significantly different from the hydrogen atom. Let’s look at these energies.
Electron Energies in Many-Electron Atoms
In H, electron energy depends only on principal quantum number n. Therefore, the 3s, 3p, and 3d atomic orbitals all have the same energy.
However, in many-electron atoms, the orbital energies depend on l, in addition to n.

Z is the effective nuclear charge, which depends on l. It is reduced from the full charge of the nucleus because of a phenomenon known as shielding by other electrons in the atom.
The shielding effect is the decrease in nuclear attraction on an electron, caused by the presence of other electrons in underlying orbitals, and due to electron-electron repulsion.
The effective nuclear charge is equal to the actual nuclear charge subtracted by the shielding effects, or mathematically:
Effective nuclear charge = actual nuclear charge – shielding effects
So, while the effective nuclear charge of H is 1.00, the effective nuclear charge for He is less than 2.
The greater the effective nuclear charge, the lower the electron energy. A larger nuclear charge exerts a stronger attractive force which draws the electron closer and lowers its energy.

This is a diagram of the radial distribution function, and explains why the electron’s energy depends on l. We see that the trend in orbital penetration is s>p>d>f.
Electron spin in Many-Electron Atoms
Spin is important in many-electron rules, as we will see in the rules governing atomic orbitals below. We can imagine the two values of ms (+1/2 and -1/2) as corresponding to an electron spinning clockwise and anti-clockwise.
Pauli Exclusion Principle
The Pauli Exclusion Principle explains why all the electrons in a many-electron atom don’t end up in the 1s orbital. It states that:
- No two electrons in an atom can have the same values for all four quantum numbers, or in other words:
- Only two electrons can be in one orbital (same n, l, and ml) and these electrons must have opposite spins (i.e. paired).
Hund’s Rule
According to Hund’s rule, the groundstate corresponds to electrons occupying the orbitals of a subshell singly and with parallel spins until each orbital has one electron.
If there are electrons in the same orbital, they will repel each other, and will have a higher energy state. Also note that this rule only applies to orbitals that have the same energy.
Electron Configurations
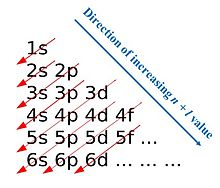
The distribution of electrons in the orbitals of many-electron atoms is represented by an electron configuration. All systems try to attain the lowest possible energy state. The Aufbau Principle involves filling the lowest energy orbital first, taking into account the Pauli Exclusion Principle and Hund’s rule.
The orbital filling order is: 1s<2s<2p<3s<3p<4s<3d<4p…
The ground state configuration gives the minimum energy to the atom as a whole. So, some examples of ground state configurations, along with the orbital diagrams on the right, are as follows:
Noble gas cores can also be used to abbreviate electron configurations.
There are exceptions to the filling order. In the d orbitals, the numbers 5 and 10 have a special stability, even though they may come later in the filling order.
Electron Configuration of Monoatomic Ions
When forming cations (positive ions), electrons are removed in order of decreasing n.
When forming anions (negative ions), the filling order is the same.
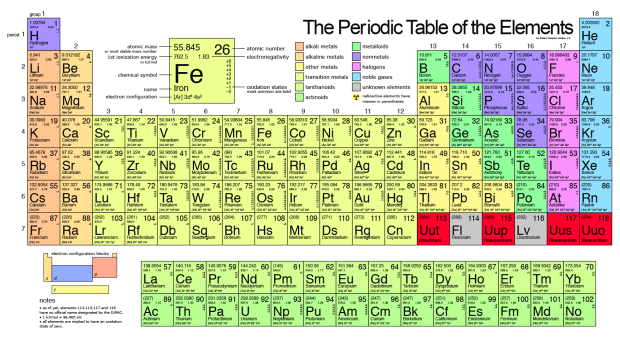
The Periodic Table
When the elements are arranged according to atomic number, we can see a periodicity in chemical and physical properties. (Bonus fact: Mendeleev discovered this!)
Groups are the vertical columns that are families of elements, and have similar chemical properties.
Periods are the horizontal rows in the table.
For example, group 1 form the alkali metals (except H), group 2 form the alkali-earth metals, group 17 form the halogens, and group 18 form the noble gases.
Transition Metals
The elements for which the last electron enters the d orbital are the d block elements, which include all the transition metal elements and the elements in group 2B.
The 3d orbital for Z=19 and Z=20 is above the 4s orbital, while the 3d orbital actually lies below the 4s orbital for Z above 20. So, V (Z=23) 3d^3 4s^2, for example. Because the most probable distance of the 3d electron from the nucleus is less than that for a 4s electron, two 3d electrons repel each other more strongly, hence the electron configuration for V^7.
In transition metals, the 4s electrons are lost first.
Inner Transition Metals
These are known as the f block elements because in most cases the last electron enters to f orbital. The first series is known as Lanthanides (rare earth elements) and the second is the Actinides.
Valence Electrons
Electrons in the outermost electron shell (highest n, i.e. valence shell) are called valence electrons. Group properties are due to having the similar valence shells. The inner electrons, aka core electrons, don’t play an important role in chemical properties of the atom.
Periodic Atomic Properties
Periodic trends include atomic size, ionization energy, and electron affinity. Let’s get to some of them below.
1. Atomic Size
The atomic radius is defined to be one half the distance between the centers of identical touching atoms (when the attractive forces between atoms are balanced by repulsive forces). We use x-ray diffraction in solids and rotational spectroscopy in gases to determine atomic radii.
(There’s three types of atomic radii: the metalllic radius, the covalent radius, and the van der Waal radius, but that’s not really important for what we want to learn here.)
Atomic radii increase from top to bottom within a group on the periodic table because each new period starts a new shell which has an electron density peaked at a distance further from the nucleus.
The atomic radii decrease from left to right across a period on the periodic table because the nuclear charge increases across a period, and since the added electrons go into the same shell, the shielding effect doesn’t increase.
Ionic Radii
Cations (+) are smaller than parent atoms because the same nuclear charge now attracts fewer electrons.
Anions (-) are larger than parent atoms because of the increased repulsion between electrons.
2. Ionization Energy (IE)
The ionization energy is defined as the minimum energy required to remove an electron from a gaseous ground-state atom or ion.
The first ionization energy is the amount of energy required to remove the most loosely bound electron from the valence shell.
For a particular element, each successive ionization energy is higher than the previous, so that electrons below the valence shell will have very high IE.
IE decreases from top to bottom within a group because (1) valence electrons are physically further from the nucleus; (2) more core electrons give higher shielding effect.
IE increases from left to right across periods because atomic size decreases and effective nuclear charge increases.
However, there are exceptions. For example, boron has a lower IE than beryllium because the lone p electron on B is easier to remove than a s electron from a filled, stable subshell on Be.
Some conclusions:
- Because of the low IE, elements on the left side of the table form positive ions easily and are known as metals.
- Elements on the upper right corner are non-metals because they have large IE.
- IE is important for understanding ionic chemical bonding. (Definition of electronegativity involves IE.)
3. Electron Affinity (EA)
The electron affinity is the energy released upon the addition of an electron to a gaseous ground-state atom or ion.
Trends are not really present with EA, except for this: elements in the top right corner tend to have the largest EA (especially around the halogens).
EA is an important concept in bonding.
Well, thank goodness we’re at the end! It’s been a long day for me, but I hope I’ve imbued some knowledge onto someone at the end of the day (literally). We’ve learned about electron energies, electron configurations for many-electron atoms, and the ordering and trends of the periodic table.
Come back regularly for more posts, and as usual, leave any questions and feedback in the comment section, I will get back to you soon!
Lots of love,


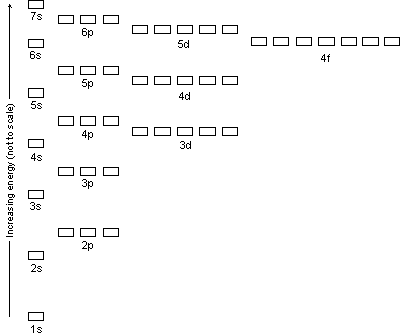
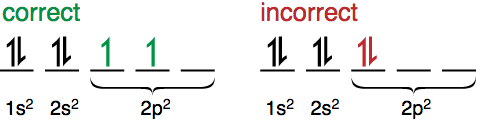


One thought on “A look at “many-electron atoms””